However, this ban on testing cosmetics on animals leaves animal testing for biomedical research as laws and regulatory agencies worldwide require that medicine and medical devices are tested on animals before clinical trials on humans. In South Korea, the Asan Institute for Life Sciences (AILS) is the largest and most advanced biomedical science and engineering research institute with over 500 MDs and nearly 800 research experts. At AILS, Dr. Ho-Young Song’s translational research laboratory develops devices and drugs tested on animals before moving onto humans to try to improve health outcomes. Translational research is the process of applying knowledge gained from clinical trials to techniques and tools that address critical medical needs. According to Song, “Experiments on animals are necessary to advance medical and biological knowledge. The effects of the substances tested are the same on man as on animals and this gives scientists that best opportunity to develop safe and effective products for the health of humans.”
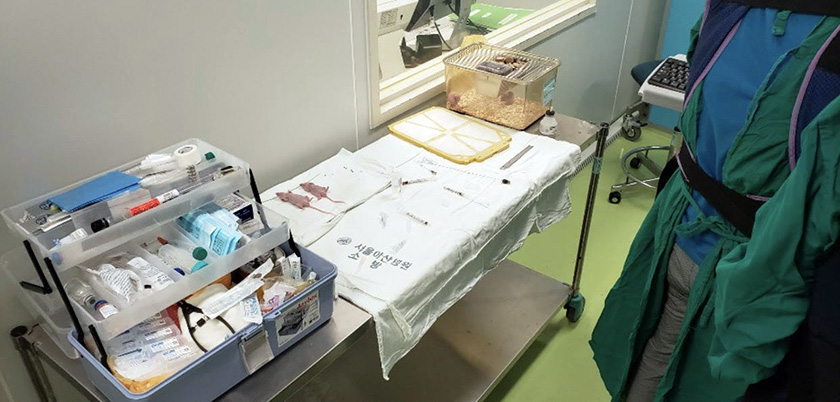
Song explained his area of expertise, which is the use of stents in interventional radiology. “For my research, stents are placed in mice mainly for esophagus and airway obstruction that would eventually aid patients who had surgically untreatable lesions. Also, stents are inserted in a minimally invasive procedure used to improve blood flow when a vein or artery is too narrow or blocked.” The use of stents in mice allows for the investigation of the response to stent injury.
Direct observation of an experiment involving two mice being inserted with a stent showed the rigorous government regulations that are in place to guarantee that the animal subjects are treated humanely and responsibly. The guidelines set high standards of care for lab animals with regard to their housing, feeding, cleanliness, ventilation and medical needs. The use of anesthesia was required with the mice for the potentially painful procedure and during post-operative care. The researchers are also required to justify their need for animals, select the most appropriate species, and use the fewest number of animals possible to answer a specific research question. AILS is required to have at least one veterinarian and one community representative who are not affiliated with the institution, to oversee the well-being of the animals. The animal testing industry has also gone beyond government regulations by creating the 3Rs campaign, which advocates the search for the replacement of animals with non-living models; reduction in the use of animals; and refinement of animal use practices.
Opponents of animal testing for biomedical purposes take the stance that animal testing should be banned because it is unethical and the results do not guarantee the safety of the products. While their arguments should be respected and listened to, the total elimination of animal testing would significantly set back the development of essential medical devices and medicines that save the lives of humans.

Yunho Choi
Junior (Grade 11)
Seoul Scholars International